Sanford stem cell trial crosses key threshold in offering orthopedic treatment
Dec. 21, 2018
This paid piece is sponsored by South Dakota Biotech.
Sanford Research has hit a key milestone in its efforts to gain FDA approval for ground-breaking use of stem cells in orthopedics and is already pursuing new trials to broaden its efforts.
Early data has been released showing that the first patients who were part of a clinical trial using adipose-derived stem cells to treat rotator cuff tears had no harmful effects from the treatment.
“We know all our patients were safe, but a significant amount of our patients also had pain relief and some function relief,” said Tiffany Facile, Sanford’s director of regenerative medicine.
“That really contributes to the next phase.”
The next step – a phase two trial that is intended to show the treatment’s efficacy – will include about 200 patients at 10 sites, starting in early 2019.
“That study duration is about a year,” Facile said.
Sanford has worked in partnership with a hospital in Munich, Germany, for years as the treatment was pioneered and offered to patients there. So while the results in the U.S. are welcomed, they’re not entirely surprising, she said.
“We know from patients in Munich that there is relief. This is a confirmation of what we knew, but there’s additional excitement because this could provide another treatment option for patients here, and it means a lot to me to be able to tell that to patients.”
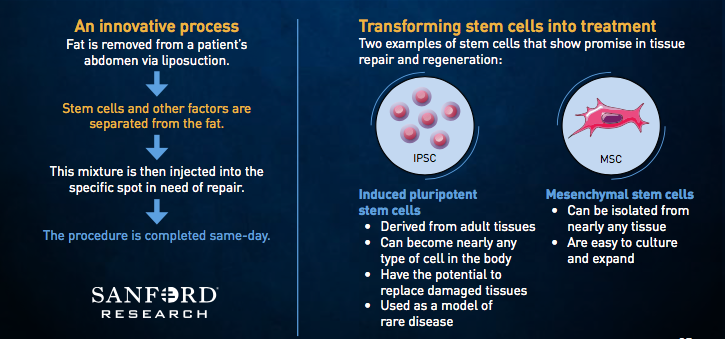
At the same time, Sanford is working through studies using adipose-derived stem cells to treat osteoarthritis in the wrist and in the back. Like the rotator cuff study, researchers have started by testing for safety and will follow with a trial for efficacy.
“Sanford Health is well connected nationally and internationally in the regenerative medicine world,” Facile said. “I think because we have set the standard and maintained a great relationship with the FDA, we will continue to lead in this space.”
Facile and David Pearce, executive vice president of innovation and research, will present in early 2019 at the World Stem Cell Summit in Miami, which features global leaders in the stem cell and regenerative medicine community.
“Sanford is really trying to lead and be a good mentor for others to work with the FDA,” Facile said. “We can do it. We can do it the right way, but you just have to trust in the science and have science in the clinical application. We are sharing our story about how it can be done in the best way, and it’s an extreme honor.”
Sanford’s leadership in regenerative medicine is a huge asset in growing South Dakota’s visibility within the bioscience industry, said Joni Johnson, executive director of South Dakota Biotech.
“This sort of activity in clinical trials is so exciting for our state,” Johnson said. “It broadens our relationships, leads to additional collaboration and helps continue to attract the sort of research talent that is lifting up our entire bioscience economy.”
Sanford’s success in pursuing stem cell clinical trials has bolstered its ability to recruit and collaborate, Facile agreed.
“It’s exciting. This is a competitive field, and we’re meeting with scientists nationally who are looking for clinical sites to conduct their studies, and Sanford is that place. That’s Sanford. So it gets me excited, and the fact that we’re following the FDA and bringing this treatment to patients in the right way is so important to take into consideration. It’s the right thing for the patient.”








